Ari R Youderian, MD
Read complete study: Benefit of intraoperative navigation on glenoid component positioning during total shoulder arthroplasty
I have now performed my first 40 shoulder replacement cases with computer assisted orthopaedic surgery (CAOS). As an early adopter of this technology, the meta-analysis by Sadoghi et al, “Benefit of intraoperative navigation on glenoid component positioning during total shoulder arthroplasty” was very intriguing to me. The authors reveal seven cases of comparative studies between CAOS and standard shoulder replacement. The biggest finding was a combined 6 degree difference in glenoid version, but they found limited data and differences in inclination or other parameters. The study emphasizes the point that navigation will continue to demonstrate improved accuracy in postoperative position, but we are only scratching the surface with the power of this technology. As a fairly high volume shoulder surgeon, even I can easily see a difference in glenoid placement, angulation and screw positions as I compare my postoperative radiographs from my navigated cases to my pre-navigation cases.
The good news is that CAOS for shoulder replacement is finally here. This is not the same as patient specific instrumentation; it’s a step further. The newest CAOS system allows for robust planning, continuous feedback to the surgeon throughout the case and the ability to deviate from the plan but still always know where you are. The authors make a great point about the added benefit of both accuracy and reliability. Not only do these systems allow you to place a glenoid within 1mm and degree of your plan, but they will commonly decrease the margin of error. This is a common theme of eliminating outliers, seen with the knee CAOS systems, especially for those who perform these cases less often.
During my fellowship, CAOS was not available. My mentor was finishing up his work on the first patient specific navigation system, and we used a robust 3-D planning tool. I quickly bridged the gap between the standard radiograph and 2-dimensional planning seen in residency to the eye-opening concepts of 3-D, including planning and implementation of glenoid sizing, seating, use of augmented implants, finding the best bone for fixation and bone graft planning in severe deformity cases.
Preoperative planning software has allowed me to more accurately choose glenoid sizes and augmentation, as well as estimate the patient’s native glenoid position.
Since that time, these concepts have permeated in each case that I carefully planned, but I did not have the tools to translate them to the operating room. Previous studies have demonstrated that the use of preoperative planning software alone adds to the accuracy of glenoid placement. Preoperative planning software has allowed me to more accurately choose glenoid sizes and augmentation, as well as estimate the patient’s native glenoid position. In addition, augmented glenoids are much easier to place with CAOS, as the guessing of how much bone to remove and at what angle to start are removed from the equation. The majority of my decision making is now performed prior to the start of the surgery, and it is then executed more promptly and efficiently during the surgery.
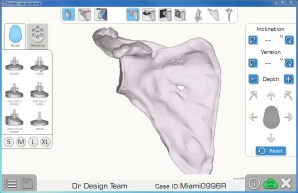
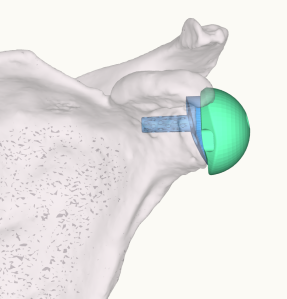
One important clarification that this study does not address is the difference between cadaver studies, virtual studies and in vivo studies.
I don’t believe we have been able to prove anything yet in terms of outcome studies because the technology is brand new. The majority of the studies in this article are cadaver-based studies. After performing both cadaver studies and real patient cases, there is a stark difference between the two. Cadaver studies likely minimize the radiographic differences in all planes and these differences would likely be larger, statistically and clinically relevant, in live patients and in patients with more severe deformities. Cadaver studies do not present the soft tissue restraints and tension found in live surgery. We are often more delicate with soft tissue releases, limit excision of tissues, and have both internal and external restraints that would add to the mal-alignment in real patients. Nonetheless, there is a void in larger outcome studies, both radiographic (postoperative CT scans would be the most accurate) and clinical, that are at least two to three years away.
This study does help point out some of the areas of future investigation. One of the most important findings from using this technology is that there is still a lot we do not know. CAOS will add to the data and our ability to improve both outcomes and longevity of implants. It is not uncommon to see community surgeons perform severe errors in position that lead to failure. In addition, while we have good outcomes overall in shoulder replacement, we must strive to improve these results and improve the duration of this success. There is more to glenoid fixation than glenoid version as well. This includes optimizing everything from glenoid seating and correcting version to the patient’s premorbid version (not necessarily zero) to minimizing medialization and subchondral bone loss.
This study only briefly addresses the reverse shoulder replacement. For the reverse shoulder, I agree that the benefits of accurate glenoid positioning can be extrapolated to the reverse shoulder. Fixation of the baseplate is obviously critical. But as we have improved the general fixation techniques of the reverse baseplate, it may be just as important to focus on additional factors. With the CAOS systems, by placing the baseplate and sphere accurately to plan, we can also minimize notching, optimize range of motion and impingement, place screws into good bone stock, avoid neurovascular structures, and improve our indications for use of augmentation to correct deformities.
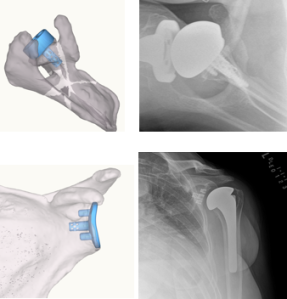 There is no doubt that after using this technology, it adds to my acuity in the operating room. I recognize bone deformity much more easily and question my position and choice of implant much less during the surgery. It has optimized my time in performing the operation, has cut down on my time in the more complicated cases, and reduced the time needed for setup. This study sheds light on the fact that shoulder navigation is all very new. Our data set and proof of value will continue to grow as we use it more.
There is no doubt that after using this technology, it adds to my acuity in the operating room. I recognize bone deformity much more easily and question my position and choice of implant much less during the surgery. It has optimized my time in performing the operation, has cut down on my time in the more complicated cases, and reduced the time needed for setup. This study sheds light on the fact that shoulder navigation is all very new. Our data set and proof of value will continue to grow as we use it more.
Ari Youderian, MD, is a board-certified orthopaedic surgeon specializing in complex upper extremity injuries. Dr. Youderian received his medical degree and completed his residency at the University of Illinois at Chicago College of Medicine. He then completed a fellowship in shoulder and elbow at the Cleveland Clinic.
